SUTD researchers develop low-cost electroporation device that could expand global access to cancer drugs
SUTD - Denise Lee, Sophia S. Y. Chan, Nemanja Aksic, Natasa Bajalovic, and Desmond K. Loke
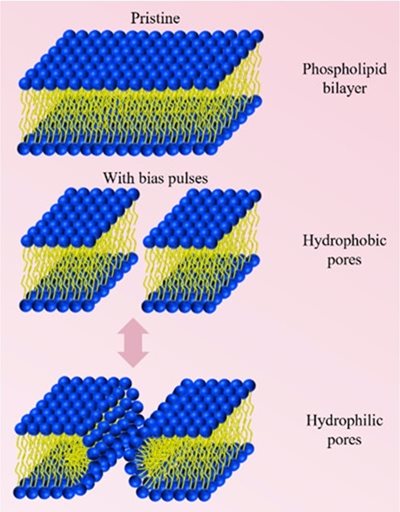 Schematic of the process of pore formation in phospholipid bilayer after electroporation.
Schematic of the process of pore formation in phospholipid bilayer after electroporation.
Researchers from the Singapore University of Technology and Design (SUTD) have developed a microsize-gap multiple-shot electroporation (M2E) device that could improve the effectiveness of delivering cancer drugs at a lower cost, globally.
“One of the goals of the scientific community is to develop a method for cancer drug delivery that is simple, manufacturable and low-cost,” said Desmond Loke, an assistant professor at SUTD and principal investigator of this research.
A way to enhance prospects for cancer drug delivery may be through electroporation—a method in which a very weak electric pulse is applied to cells to temporarily open holes in their membranes. The cancer medicines can be sent in through these holes. The application of electroporation together with cancer drugs could enhance both drug effectiveness and accessibility.
The researchers integrated transparent electrodes into the device to enhance visualisation of cancer drugs. “The narrow gap between electrodes allow us to achieve sufficiently strong electric field using a few volts rather than several tens of volts applied in traditional electroporation,” said Assistant Prof Loke. This low voltage, plus electrode transparency, minimise energy consumption and facilitate visibility, which help avoid unsafe usage of the drugs and limited imaging of drug transport during drug testing–both of which are common problems of traditional electroporation systems.
Additionally, in electrochemotherapy applications, tumour cells can be permeabilised by electroporation, thereby enhancing their uptake of chemotherapeutic drugs such as bleomycin and cisplatin.
The researchers tested the M2E device using cancer-drug-related molecules. The device allows cancer cells to show a time window for the uptake of molecules of 2 hours, which is 400% larger than conventional electroporation systems. Furthermore, it is reusable. The researchers suggest that the M2E system could work with associated drugs for treating Covid-19 as well.
“The device developed by SUTD doesn’t require specialised components, expensive materials or a complicated fabrication process, unlike traditional electroporation machines,” Assistant Prof Loke noted.
Optimisations of the M2E system are currently underway and researchers expect that it could take a few years for the device to complete clinical study and be ready for wider use. “We hope the M2E system could provide a path to significantly improve the cancer drug delivery process and allow a more uniform distribution of cancer drugs to under-resourced and underserved regions worldwide,” added Assistant Prof Loke.
The other researchers involved in this work were Denise Lee, Sophia Chan, Nemanja Aksic and Natasa Bajalovic, who are also from SUTD. This work was published in the journal American Chemical Society (ACS) Omega.
Acknowledgements:
We thank J. S. Naikar, W. C. Teoh, and L. T. Ng for inspiring discussions. This work was supported by the Ministry of Education, Singapore (MOE2017-T2-2-064), MIT-SUTD International Design Centre, Changi General Hospital-SUTD, Singapore (CGH-SUTD-HTIF2019-001), SUTD-Zhejiang-University (SUTD-ZJU (VP) 201903), and National Supercomputing Centre, Singapore (15001618) grant programs. D. Lee, S. S.-Y. Chan, and N. Aksic thank the Ministry of Education and SUTD for the scholarship support.
Reference:
Ultralong-Time Recovery and Low-Voltage Electroporation for Biological Cell Monitoring Enabled by a Microsized Multipulse Framework, ACS Omega. (DOI: 10.1021/acsomega.1c04257)